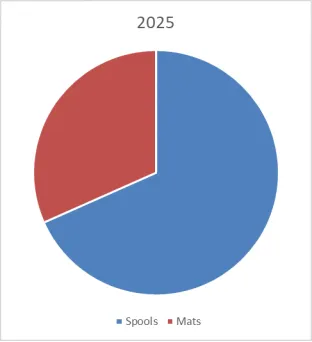
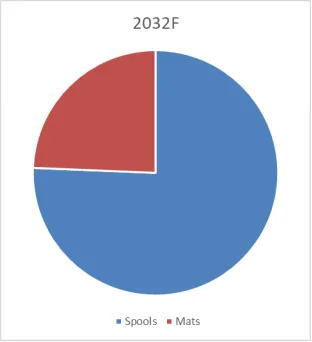
Porous Hard Carbon Market Summary
Porous Hard Carbon is an advanced anode material designed for lithium-ion and sodium-ion batteries, characterized by a highly developed porous structure formed through controlled carbonization and microstructure engineering of carbon precursors. The interconnected pore network enhances ion storage and transport efficiency, enabling high capacity and stable cycling performance in energy storage systems. Its advantages include high specific capacity, strong structural stability, and long battery service life.
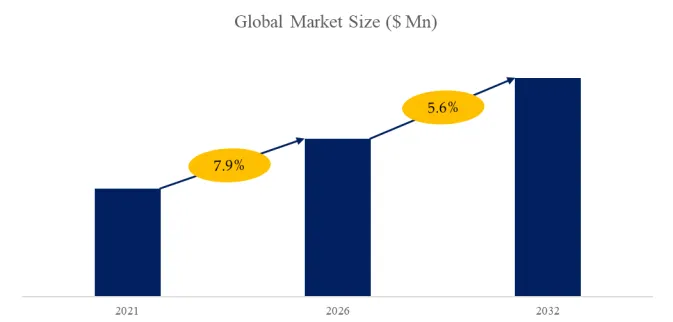
According to the new market research report “Global Porous Hard Carbon Market Report 2026-2032”, published by QYResearch, the global Porous Hard Carbon market size is projected to reach USD 10.59 billion by 2032, at a CAGR of 35.0% during the forecast period.
Figure00001. Global Porous Hard Carbon Market Size (US$ Million), 2021-2032
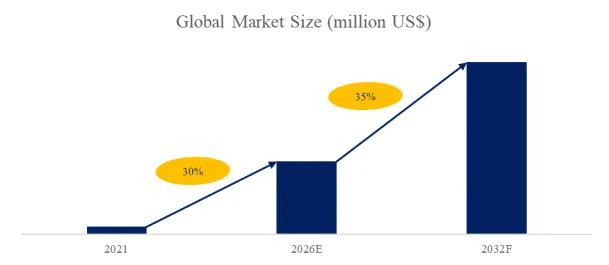
Above data is based on report from QYResearch: Global Porous Hard Carbon Market Report 2026-2032 (published in 2026). If you need the latest data, plaese contact QYResearch.
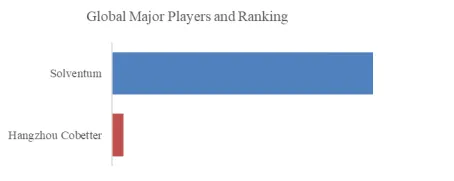
Figure00002. Global Porous Hard Carbon Top 15 Players Ranking and Market Share (Ranking is based on the revenue of 2025, continually updated)
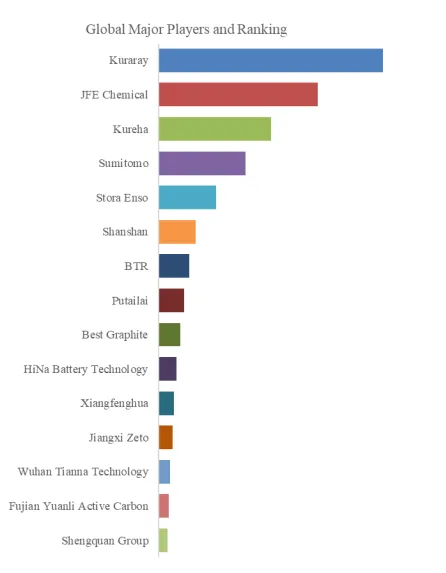
Above data is based on report from QYResearch: Global Porous Hard Carbon Market Report 2026-2032 (published in 2026). If you need the latest data, plaese contact QYResearch.
According to QYResearch Top Players Research Center, the global key manufacturers of Porous Hard Carbon include Kuraray, JFE Chemical, Kureha, Sumitomo, Stora Enso, Shanshan, BTR, Putailai, Best Graphite, HiNa Battery Technology, etc. In 2025, the global top 10 players had a share approximately 70.0% in terms of revenue.
Industrial Chain
The upstream of Porous Hard Carbon primarily utilizes high-carbon raw materials as feedstock, forming the foundational basis of the production chain. Key raw materials include coconut shells and asphalt, which determine the initial carbon structure and impurity levels. Leading suppliers in this segment include Olam Group, Cargill, Baowu Carbon Materials, and Sinopec. The quality, consistency, and supply stability of these materials directly affect midstream processing efficiency and the electrochemical performance of the final porous hard carbon.
The midstream stage is the core of technical value creation, focusing on carbonization, pore structure engineering, particle size control, and purity optimization. Manufacturers refine high-carbon raw materials into porous hard carbon with uniform electrochemical properties suitable for battery applications. Precise control over microstructure and surface characteristics ensures consistent performance, energy density, and cycle life in downstream battery systems. Strong process management and quality control capabilities are essential to maintain reproducibility and meet high performance standards.
Porous Hard Carbon is mainly applied as an anode material in lithium-ion and sodium-ion batteries, serving as a critical component in energy storage systems. Key downstream customers include Panasonic, Samsung SDI, CATL, BYD, and Farasis Energy. Material consistency, purity, and particle structure directly affect battery efficiency, lifespan, and safety, making midstream-to-downstream integration a key determinant of industry competitiveness.
Influencing Factors
Key Drivers:
The growth of Porous Hard Carbon is primarily driven by fast-charging demands in both consumer electronics and electric vehicles. In consumer electronics, smartphones, tablets, and laptops require rapid charging and long cycle life, driving the need for high-rate anode materials. In electric vehicles, high-power fast charging and extended driving range demand low-polarization, high ion-transport capacity anodes. Porous hard carbon, with its micro-porous structure, high surface area, and excellent cycle stability, can meet the fast charge–discharge and long-life requirements in both sectors, making it a key material for fast-charging applications.
Challenges:
The industry faces challenges from raw material price fluctuations, complex manufacturing processes, and strict consistency requirements. In fast-charging applications, whether for consumer electronics or EV batteries, precise control of pore structure, conductivity, and particle distribution increases midstream processing difficulty and raises quality management costs. Downstream customers also impose high standards for safety, stability, and cycle life, further increasing market entry barriers.
Industry Trends:
Looking ahead, porous hard carbon is expected to evolve toward higher specific surface area, improved conductivity, and optimized ion transport to meet the dual demands of consumer electronics and EV fast charging. Pore structure engineering, particle size optimization, and surface modification will enhance performance under high-rate charge–discharge conditions. Industry competition will increasingly focus on fast-charging performance, cycle life, and material consistency, favoring companies with advanced processing capabilities and reliable supply chains.
About The Authors
Lead Author: Julie Zhang
Email: zhangjianan@qyresearch.com
Julie Zhang, a key industry analyst a industry analyst of QYResearch (Beijing Hengzhou Bozhi International Information Consulting Co.,Ltd.), focuses on market research and trend forecasting of the entire industry chain upstream and downstream of the electric vehicle and lithium battery industry, we are good at providing strategic market insights through in-depth data mining, focusing on trends and technological innovations in the automotive and lithium battery industry, and helping the company achieve sustainable success in the highly competitive market environment. Typical studies include Electronic Fusing IC, EV Skateboard Platform, Electric Vehicle Controller, Automotive Interior Monitoring System, Automotive PCIe Switch Chips, End-To-End Automotive Software Platform, LiFSI Electrolyte Salts, Portable Power Supply, Outdoor Mobile Powers, and Solar Energy Storage Battery, etc.
About QYResearch
QYResearch founded in California, USA in 2007. It is a leading global market research and consulting company. With over 17 years’ experience and professional research team in various cities over the world QY Research focuses on management consulting, database and seminar services, IPO consulting (data is widely cited in prospectuses, annual reports and presentations), industry chain research and customized research to help our clients in providing non-linear revenue model and make them successful. We are globally recognized for our expansive portfolio of services, good corporate citizenship, and our strong commitment to sustainability. Up to now, we have cooperated with more than 60,000 clients across five continents. Let’s work closely with you and build a bold and better future.
QYResearch is a world-renowned large-scale consulting company. The industry covers various high-tech industry chain market segments, spanning the semiconductor industry chain (semiconductor equipment and parts, semiconductor materials, ICs, Foundry, packaging and testing, discrete devices, sensors, optoelectronic devices), photovoltaic industry chain (equipment, cells, modules, auxiliary material brackets, inverters, power station terminals), new energy automobile industry chain (batteries and materials, auto parts, batteries, motors, electronic control, automotive semiconductors, etc.), communication industry chain (communication system equipment, terminal equipment, electronic components, RF front-end, optical modules, 4G/5G/6G, broadband, IoT, digital economy, AI), advanced materials industry Chain (metal materials, polymer materials, ceramic materials, nano materials, etc.), machinery manufacturing industry chain (CNC machine tools, construction machinery, electrical machinery, 3C automation, industrial robots, lasers, industrial control, drones), food, beverages and pharmaceuticals, medical equipment, agriculture, etc.
About Us:
QYResearch founded in California, USA in 2007, which is a leading global market research and consulting company. Our primary business include market research reports, custom reports, commissioned research, IPO consultancy, business plans, etc. With over 18 years of experience and a dedicated research team, we are well placed to provide useful information and data for your business, and we have established offices in 7 countries (include United States, Germany, Switzerland, Japan, Korea, China and India) and business partners in over 30 countries. We have provided industrial information services to more than 60,000 companies in over the world.
Contact Us:
If you have any queries regarding this report or if you would like further information, please contact us:
QY Research Inc.
Add: 17890 Castleton Street Suite 369 City of Industry CA 91748 United States
EN: https://www.qyresearch.com
Email: global@qyresearch.com
Tel: 001-626-842-1666(US)
JP: https://www.qyresearch.co.jp