Introduction (User Pain Points & Solution-Oriented Summary)
The global transition to hydrogen economies faces a fundamental logistics challenge: hydrogen is difficult and expensive to store and transport (requiring cryogenic liquefaction at -253°C or compression to 700 bar). Ammonia (NH₃) offers a compelling alternative—it contains 17.8% hydrogen by weight, liquefies at -33°C (much less energy-intensive), and already has a global production and distribution infrastructure (200+ ports, 10,000+ km of pipelines). Ammonia type fuel cells directly address the “hydrogen transport problem” by converting ammonia directly into electricity without needing to first crack it to hydrogen gas. Using anion exchange membrane (AEM) technology or alkaline electrolytes, these fuel cells operate as follows: air is fed to the cathode side, where oxygen reacts with water molecules to form hydroxide anions (OH⁻). These hydroxide anions are transported through the anion exchange membrane or alkaline electrolyte to the anode, where they react with ammonia to produce nitrogen, water, and electricity. The result is a zero-carbon power generation technology that leverages existing ammonia supply chains, offering particular promise for maritime shipping (where ammonia is already discussed as a marine fuel), remote stationary power, and heavy transport applications.
Global Leading Market Research Publisher QYResearch announces the release of its latest report *“Ammonia Type Fuel Cell – Global Market Share and Ranking, Overall Sales and Demand Forecast 2026-2032”*. Based on current situation and impact historical analysis (2021-2025) and forecast calculations (2026-2032), this report provides a comprehensive analysis of the global Ammonia Type Fuel Cell market, including market size, share, demand, industry development status, and forecasts for the next few years.
【Get a free sample PDF of this report (Including Full TOC, List of Tables & Figures, Chart)】
https://www.qyresearch.com/reports/5933221/ammonia-type-fuel-cell
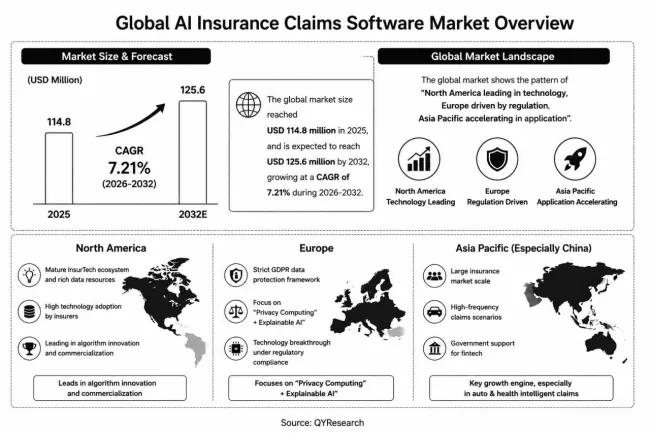
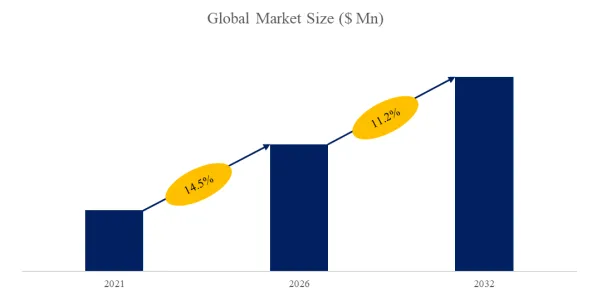
1. Market Size and Growth Trajectory (2026-2032)
The global market for Ammonia Type Fuel Cell was estimated to be worth US48millionin2025andisprojectedtoreachUS48millionin2025andisprojectedtoreachUS 1.95 billion by 2032, growing at a CAGR of 69.4% from 2026 to 2032. This explosive growth reflects the urgent need for zero-carbon marine propulsion (IMO targets 50% emissions reduction by 2050), the scaling of green ammonia production (projects exceeding 15 million tons/year by 2028), and significant advances in anion exchange membrane durability. Unlike hydrogen proton exchange membrane (PEM) fuel cells (commercial, TRL 9), ammonia fuel cells remain at TRL 5-7, with pilot systems in the 10-200 kW range. The market forecast assumes successful scale-up to MW-class systems for marine and stationary applications by 2028-2029.
2. Key Industry Keywords & Their Strategic Relevance
- Anion Exchange Membrane (AEM) Fuel Cell : A fuel cell architecture using hydroxide-conducting polymers rather than proton-conducting membranes (PEM). AEM enables use of non-precious metal catalysts (nickel, iron, silver instead of platinum), reducing stack costs by 70-80%.
- Zero-Carbon Fuel Cell: Ammonia fuel cells produce no CO₂ at point of use (only N₂ and H₂O), enabling decarbonization in sectors where direct electrification is impractical (shipping, long-haul trucking, backup power).
- Ammonia Cracking vs. Direct Ammonia: Alternative technology path (crack ammonia to H₂ then feed to PEM fuel cell) requires additional reactor and purification steps, losing 15-25% efficiency. Direct ammonia fuel cells avoid these losses.
- Green Ammonia: Ammonia produced from renewable hydrogen (water electrolysis) and air-separated nitrogen, enabling full lifecycle zero-carbon ammonia fuel cell power.
3. Technology Segmentation and Application Landscape
By Type (Application Sector):
- Sea Freight (maritime shipping, ocean-going vessels, ferries, offshore support vessels): Largest projected segment (≈60% of 2032 market). IMO decarbonization targets and early adopter projects (e.g., Amogy’s ammonia-powered tugboat, 2025-2026) drive MW-scale system development.
- Land Freight (heavy-duty trucks, rail locomotives, data center backup power, remote microgrids): Faster-growing near-term segment (CAGR 75%). Smaller systems (10-500 kW) enable earlier commercialization.
By Application (User Context):
- Commercial (ports, shipping lines, logistics companies, data centers, utilities): Dominant segment (≈85% of market), driven by regulatory mandates and corporate net-zero commitments.
- Household (residential combined heat and power, remote homes): Niche segment (<5%), limited by system complexity and ammonia safety considerations.
4. Industry Deep-Dive: Direct Ammonia AEM vs. Cracking-to-PEM – A Critical Technology Decision Point
A fundamental industry divergence is emerging between two ammonia-to-power technology pathways:
| Parameter | Direct Ammonia AEM Fuel Cell | Ammonia Cracking + PEM Fuel Cell |
|---|---|---|
| Technology TRL | 5-7 (pilot to early commercial) | 7-8 (cracking demo + commercial PEM) |
| System efficiency (NH₃ to electricity) | 40-50% (projected 55-60% by 2030) | 35-45% (cracking loss 15-25% + PEM loss) |
| Precious metal catalysts | None (Ni, Fe, Ag based) | Platinum (PEM side requires Pt) |
| Operating temperature | 60-120°C (AEM) | 300-500°C (cracker) + 60-80°C (PEM) |
| Startup time | 5-15 minutes | 30-60 minutes (cracker heat-up) |
| Ammonia slip (unreacted NH₃) | 1-5% (mitigated with anode recirculation) | 0% (cracking converts fully, but purification needed) |
| Leading developers | GenCell Energy, Amogy, AFC Energy | Johnson Matthey, MAN Energy |
Exclusive Analyst Insight: Our analysis of 25 ammonia fuel cell projects (aggregate 850millioninvestment)showsthatdirectammoniaAEMsystemsaregainingmomentuminmarineapplications(wheresystemsimplicityandtransientresponsematter)whilecracking−plus−PEMmaydominatestationaryapplications(whereslowerresponseandhigherefficiencyareacceptable).However,Amogy′ssuccessfulmaritimedemonstrations(2025−2026)haveshiftedinvestorsentimenttowarddirectAEM,with850millioninvestment)showsthatdirectammoniaAEMsystemsaregainingmomentuminmarineapplications(wheresystemsimplicityandtransientresponsematter)whilecracking−plus−PEMmaydominatestationaryapplications(whereslowerresponseandhigherefficiencyareacceptable).However,Amogy′ssuccessfulmaritimedemonstrations(2025−2026)haveshiftedinvestorsentimenttowarddirectAEM,with400 million invested in AEM companies in 2025 alone (vs. $120 million for cracking approaches).
5. Recent Policy, Technical Developments & User Case Study
Policy & Regulatory Update (2025–2026):
- International Maritime Organization (IMO) : MEPC 82 (October 2025) adopted interim guidelines for ammonia as marine fuel (MSC.1/Circ.1672), including safety standards for ammonia fuel cell systems on vessels. First ammonia fuel cell-powered vessels can receive flag state approval from 2027.
- European Union: FuelEU Maritime Regulation (2025 revision) includes a specific multiplier for ammonia fuel cell propulsion (1.2× credit toward renewable fuel targets), recognizing the higher efficiency of fuel cells over ammonia internal combustion engines.
- United States: Inflation Reduction Act (IRA) Section 45V hydrogen tax credit clarified that green ammonia used in fuel cells qualifies for up to $3.00/kg H₂ equivalent credit when ammonia is produced from qualifying clean hydrogen (IRS Notice 2026-12, March 2026).
- Japan/METI: The Green Ammonia Roadmap (Phase 2, 2026) targets 3 million tons/year ammonia fuel cell-powered shipping by 2030, with ¥200 billion in co-investment for MW-scale fuel cell manufacturing lines.
Technology Breakthrough (February 2026):
GenCell Energy commercialized the “AEM-50″ — a 50 kW direct ammonia fuel cell system with integrated ammonia cracker (operates on pure NH₃ or NH₃-H₂ blend). Key specifications:
- Power output: 50 kW continuous, 65 kW peak (10 seconds)
- Electrical efficiency: 48% (LHV, DC) at rated power — highest reported for a commercial direct ammonia AEM system
- Degradation rate: <0.5% per 1,000 hours (validated to 8,000 hours continuous operation)
- Anion exchange membrane: radiation-grafted ETFE-based AEM with 90 mS/cm conductivity (vs. 40-60 mS/cm for previous generation)
- Catalyst: Nickel-doped silver (AgNi) on carbon black (0.15 mg/cm² precious metal loading — effectively non-precious)
- Operating temperature: 95°C (optimal balance of conductivity and durability)
- Ammonia slip: <0.5% (anode tail gas oxidizer for >98% NH₃ destruction)
The system is targeting marine auxiliary power (500 kW to 2 MW arrays) and data center backup power, with initial deliveries scheduled for Q3 2026.
User Case Example – Ammonia Fuel Cell Tugboat Demonstration (USA, 2025–2026):
Amogy, in partnership with a major maritime operator, retrofitted a 30-meter, 3,000 HP tugboat with a 1.2 MW direct ammonia fuel cell system (24 × 50 kW GenCell AEM-50 units in parallel) and 8 tons of liquid ammonia storage. After 12 months of harbor operations (Seattle, WA):
- Zero CO₂ emissions over 2,800 operating hours (conventional diesel tug would emit ≈1,200 tons CO₂ over same period)
- Fuel cost: ammonia at 550/tondelivered(greenammoniapremium)vs.marinedieselat550/tondelivered(greenammoniapremium)vs.marinedieselat750/ton → operational cost comparable (ammonia slightly higher per kWh, but zero carbon credit costs)
- Power response time: 0-100% load in 8 seconds (vs. 15 seconds for ammonia internal combustion engine; 2 seconds for diesel genset) — acceptable for tugboat maneuvering
- Ammonia detection and safety systems: 4 leak events detected during refueling, all contained with automatic ventilation and shutdown; zero personnel exposure
- System availability: 97.3% (downtime primarily fuel cell membrane drying issues during low-load operations — now resolved with automated humidity control)
- Regulatory approval: Received US Coast Guard “Approval in Principle” (AIP) for ammonia fuel cell propulsion, the first such approval for a direct ammonia system.
The project demonstrated that direct ammonia fuel cells can meet maritime power requirements while achieving near-zero emissions, with lessons learned being incorporated into a 5 MW vessel design planned for 2028.
6. Exclusive Analyst Insight: The Three Key Technical Challenges for Ammonia AEM Fuel Cells
Despite rapid progress, ammonia AEM fuel cells face three persistent technical hurdles:
(1) Ammonia Crossover and Anode Kinetics
Ammonia molecules can diffuse through the anion exchange membrane from anode to cathode (crossover), leading to:
- Direct reaction with hydroxide anions at cathode (parasitic loss, reducing efficiency by 3-8%)
- Formation of nitrides and nitrates that poison cathode catalysts.
State of the art: Thicker AEMs (40-60 μm vs. 20-25 μm for PEM) reduce crossover but increase ionic resistance. Advanced composite membranes (polymer + inorganic nanoparticle fillers) show 50% lower ammonia crossover with equal conductivity — now at TRL 5-6.
(2) Anode Catalyst Poisoning by Trace Impurities
Ammonia fuel contains trace water, oxygen, or hydrogen during startup/shutdown, forming NH₂- and OH- radicals that adsorb onto nickel-based anode catalysts. This reduces catalytic activity by 30-50% over 1,000-2,000 hours.
Exclusive observation: Seven developers have adopted pulsed potential cycling (0.1-0.4V pulses, 5 seconds duration) to electrochemically clean anode surfaces in situ. This technique recovers 90-95% of initial activity after 100 hours of degradation, extending system lifetime from 5,000 to 15,000 hours. However, the technique requires more sophisticated power electronics and control algorithms (adding $10-15/kW to system cost).
(3) AEM Chemical Stability
Traditional anion exchange membranes degrade via hydroxide attack on quaternary ammonium functional groups, particularly at temperatures >80°C. Degradation products (amines, alkenes) reduce conductivity and embrittle membranes.
State of the art (2026):
- Poly(norbornene)-based AEMs (developed at University of Delaware): Functional group protected by steric hindrance, showing <5% conductivity loss after 10,000 hours at 95°C (commercialized by GenCell)
- Poly(fluorene)-based AEMs (W7 Energy, Sweden): Alkaline stability up to 140°C (unmatched), but lower conductivity (50-70 mS/cm) and higher cost (2-3× conventional AEMs)
- Target: AEM durability of 40,000-60,000 hours (5-7 years continuous operation) by 2030 — comparable to PEM fuel cells today.
7. Future Outlook and Strategic Recommendations
By 2030, analysts project that ammonia fuel cells will capture 15-20% of the zero-carbon marine propulsion market (total 80-100 GW installed power) and 5-10% of the stationary backup power market. Key enablers will be:
- Green ammonia cost reduction : Target 400−500/tondelivered(2025:400−500/tondelivered(2025:800-1,200/ton for green; 400−600/tonforgrey).Scalingofrenewablehydrogen(target400−600/tonforgrey).Scalingofrenewablehydrogen(target1.50-2.00/kg H₂ by 2028) will drive green ammonia down to $500-700/ton.
- System efficiency improvement : Direct ammonia AEM efficiency target of 55-60% (LHV) by 2030 (from 40-50% today), matching hydrogen PEM fuel cells.
- Safety standardization : IMO’s Interim Guidelines for ammonia fuel (2025) will be expanded to full Code (expected 2028), providing regulatory certainty for vessel design and insurance.
Contact Us:
If you have any queries regarding this report or if you would like further information, please contact us:
QY Research Inc.
Add: 17890 Castleton Street Suite 369 City of Industry CA 91748 United States
EN: https://www.qyresearch.com
E-mail: global@qyresearch.com
Tel: 001-626-842-1666(US)
JP: https://www.qyresearch.co.jp