QY Research Inc. (Global Market Report Research Publisher) announces the release of 2025 latest report “Medical Heater Cooler Device- Global Market Share and Ranking, Overall Sales and Demand Forecast 2026-2032”. Based on current situation and impact historical analysis (2020-2024) and forecast calculations (2026-2032), this report provides a comprehensive analysis of the global Medical Heater Cooler Device market, including market size, share, demand, industry development status, and forecasts for the next few years.
The global market for Medical Heater Cooler Device was estimated to be worth US$ million in 2024 and is forecast to a readjusted size of US$ million by 2031 with a CAGR of %during the forecast period 2025-2031.
【Get a free sample PDF of this report (Including Full TOC, List of Tables & Figures, Chart)】
https://www.qyresearch.com/reports/3886040/medical-heater-cooler-device
Medical Heater Cooler Device Product Introduction
A Medical Heater Cooler Device is a Class II critical medical device that precisely regulates patient body temperature, circulating blood, and intravenous fluids via closed-loop temperature‑controlled fluid (typically water or glycol) systems. It delivers heating or cooling through heat exchangers (for cardiopulmonary bypass) or warming/cooling blankets, maintaining normothermia or inducing controlled hypothermia/hyperthermia during cardiac, thoracic, transplant, and critical care surgeries to prevent hypothermia‑related complications and support organ protection.
According to the new market research report “Global Medical Heater Cooler Device Market Report 2026-2032”, published by QYResearch, the global Medical Heater Cooler Device market size is projected to reach USD 0.39 billion by 2032, at a CAGR of 4.6% during the forecast period.
Figure00001. Global Medical Heater Cooler Device Market Size (US$ Million), 2026 VS 2032
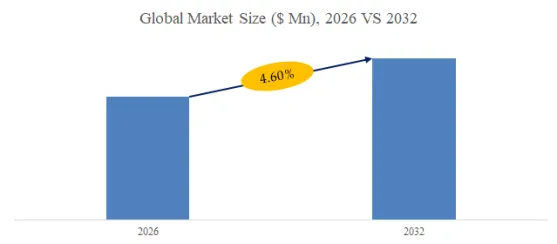
Above data is based on report from QYResearch: Global Medical Heater Cooler Device Market Report 2026-2032 (published in 2026). If you need the latest data, plaese contact QYResearch.
Figure00002. Global Medical Heater Cooler Device Top 9 Players Ranking and Market Share (Ranking is based on the revenue of 2025, continually updated)
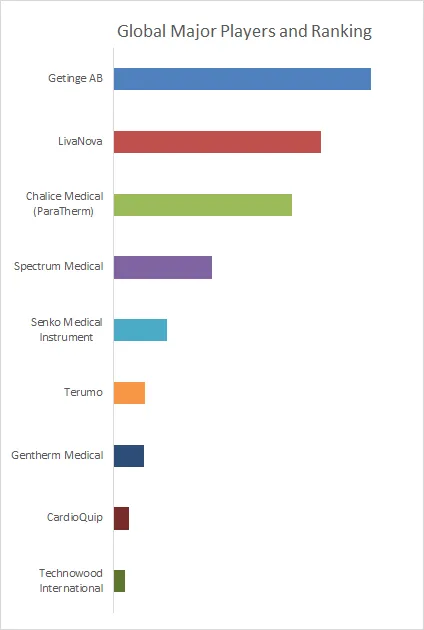
Above data is based on report from QYResearch: Global Medical Heater Cooler Device Market Report 2026-2032 (published in 2026). If you need the latest data, plaese contact QYResearch.
According to QYResearch Top Players Research Center, the global key manufacturers of Medical Heater Cooler Device include Getinge AB, LivaNova, etc. Medical Heater Cooler Device manufacturers are mainly concentrated in Europe, Japan, and North America, where strong medical-device engineering capabilities, mature cardiopulmonary surgery and perfusion ecosystems, strict quality/regulatory systems, and close hospital–industry collaboration support continuous product innovation, high reliability, and global market competitiveness.
Table 1. Medical Heater Cooler Device Industry Chain Analysis
| Item | Description | |
| Upstream | Medical-grade raw materials | Medical-grade metals (stainless steel, aluminum), biocompatible polymers, thermal fluids (sterile water/glycol blends) and fire-resistant insulation materials, all meeting ISO 10993 biocompatibility standards to ensure patient safety and prevent adverse reactions during clinical use. |
| Core thermal & control components | High-efficiency hermetic compressors or solid-state thermoelectric (Peltier) modules, precision NTC/RTD temperature sensors with ±0.1°C accuracy, closed-loop PID controllers, medical-grade peristaltic pumps with low hemolysis risk, and corrosion-resistant stainless steel heat exchangers for efficient thermal transfer. | |
| Specialized mechanical & electronic parts | IP54/IP65-rated medical enclosures, sterile single-use fluid circuit tubing, pressure relief safety valves, intuitive touchscreen control panels, isolated power supplies, and multi-level alarm systems for over-temperature, over-pressure, and flow interruption events. | |
| Midstream | Device assembly & manufacturing | Precision integration of thermal, fluid, and electronic systems; rigorous pressure leak testing; thermal performance validation across target temperature ranges (-10°C to +45°C); and adherence to ISO 13485 quality management systems for medical device production. |
| Downstream | Heart Surgery | Used in cardiopulmonary bypass (CPB) procedures to precisely regulate blood and cardioplegia solution temperatures, inducing controlled hypothermia to protect the myocardium during cardiac arrest and maintaining normothermia post-reperfusion to reduce complications. |
| Lung Surgery | Deployed in thoracic and lung transplant surgeries to manage patient core temperature, preventing perioperative hypothermia that can impair lung function and wound healing, while supporting targeted temperature management for critical care recovery. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 2. Medical Heater Cooler Device Industry Policy Analysis
| Policy | Description | |
| 1 | U.S. FDA Infection Control and Post-market Surveillance Requirements | The U.S. regulatory environment for Medical Heater Cooler Devices is heavily shaped by FDA safety communications and recommendations on water-based heater-cooler devices, requiring manufacturers and hospitals to strengthen cleaning/disinfection validation, device monitoring, adverse-event reporting, and risk mitigation for nontuberculous mycobacteria contamination, which has raised market entry and compliance thresholds but also accelerated safer product redesign. |
| 2 | EU MDR Compliance and Device Classification Regulation | In Europe, Medical Heater Cooler Devices are governed under the EU Medical Device Regulation (MDR 2017/745), with MDCG guidance specifically referencing heater-cooler units used in cardiac surgery, meaning manufacturers must meet stricter clinical evaluation, technical documentation, traceability, post-market surveillance, and notified-body review requirements, which increases regulatory costs but strengthens product standardization and international competitiveness. |
| 3 | UK MHRA Safety Alerts and Infection Reporting Framework | In the UK, MHRA drug and device alerts on heater-cooler units used in cardiac surgery require contaminated or suspected devices to be reported, removed from service when necessary, and linked infections to be notified to public health authorities, creating a policy environment that emphasizes lifecycle safety management, hospital-side vigilance, and tighter supplier quality assurance. |
| 4 | Japan PMDA / MHLW Medical Device Approval and Post-market Control | In Japan, Medical Heater Cooler Devices fall under the PMDA/MHLW medical device regulatory framework, where approval, quality management, and post-marketing safety controls are rigorous for extracorporeal circulation-related equipment, supporting high product reliability and clinical acceptance while favoring manufacturers with strong regulatory capabilities and long-term hospital partnerships. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 3. Medical Heater Cooler Device Industry Development Trends
| Development Trends | Description | |
| 1 | Miniaturization & Portable Design | Driven by demand for point-of-care, emergency, and mobile clinical use, devices are becoming smaller, lighter, and battery-powered, enabling deployment in ambulances, field hospitals, ICUs, and remote clinics while maintaining high-precision temperature control (±0.1°C accuracy) and compliance with strict medical safety standards like IEC 60601. This trend also supports easier maneuverability in crowded operating rooms and reduces the physical footprint of critical care equipment. |
| 2 | Smart & Connected Digital Integration | Increasing integration with IoT, AI-driven algorithms, and hospital clinical monitoring systems supports real-time temperature data logging, remote operation, predictive maintenance alerts, automated alarm management, and seamless EHR (Electronic Health Record) connectivity. This enhances workflow efficiency, reduces manual data entry errors, and enables data-driven perioperative temperature management to improve patient outcomes and reduce complications. |
| 3 | Advanced Thermal & Energy-Efficient Technology | Growing adoption of high-performance solid-state thermoelectric (Peltier) systems, variable-speed compressors, and optimized stainless steel heat exchangers improves energy efficiency by up to 30%, reduces noise and vibration, lowers refrigerant dependency, and extends service life, especially for high-end cardiac and critical-care applications. These technologies also minimize the risk of refrigerant leaks, enhancing patient and environmental safety. |
| 4 | Focus on Patient Safety & Infection Control | Stronger design emphasis on antibacterial materials, smooth easy-to-clean surfaces, closed sterile fluid circuits, and reduced contamination risk addresses hospital-acquired infection concerns, aligning with global clinical guidelines for cardiac surgery and critical care patient safety. Manufacturers are also integrating single-use fluid path components to further reduce cross-contamination risks between patients. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 4. Medical Heater Cooler Device Industry Development Opportunities
| Development Opportunities | Description | |
| 1 | Emerging Market Penetration & Healthcare Infrastructure Expansion | Rapid growth in healthcare infrastructure across Asia-Pacific, Latin America, and the Middle East, paired with rising rates of cardiac surgery, organ transplantation, and critical care capacity, creates strong demand for affordable, reliable heater-cooler devices. This untapped market offers manufacturers significant revenue growth, especially for mid-range, cost-effective models tailored to resource-limited clinical settings. |
| 2 | Technological Innovation & Premium Product Differentiation | Advancements in solid-state thermoelectric (Peltier) technology, AI-driven predictive temperature control, and IoT-enabled remote monitoring allow manufacturers to launch high-margin smart devices. These products address unmet clinical needs—such as real-time EHR integration and predictive maintenance—differentiating brands from commodity competitors and capturing share in premium cardiac and transplant care markets. |
| 3 | Clinical Guideline Alignment & Expanded Reimbursement Coverage | Growing clinical evidence linking perioperative temperature management to reduced surgical site infections, cardiac events, and length of hospital stay drives updated guidelines (e.g., WHO, ACC/AHA) and expanded insurance reimbursement. This policy shift incentivizes hospitals to adopt heater-cooler devices, boosting market penetration and creating stable, long-term demand for both new installations and replacement units. |
| 4 | Sustainability & Circular Economy-Driven Green Product Lines | Global healthcare sustainability mandates and eco-friendly procurement preferences create opportunities for manufacturers to develop energy-efficient, recyclable devices with modular designs. These products reduce operational costs for hospitals, align with carbon-neutrality goals, and open new revenue streams from lifecycle services like component recycling, repair, and upgrade programs. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 5. Medical Heater Cooler Device Obstacles/Challenges to Industry Development
| Obstacles/Challenges | Description | |
| 1 | Stringent Regulatory & Certification Barriers | High-cost, time-consuming regulatory pathways (e.g., FDA 510(k), CE marking) require extensive clinical testing, biocompatibility validation, and compliance with IEC 60601/ISO 13485 standards. These barriers delay market entry, increase R&D costs, and disproportionately impact small and medium-sized manufacturers, limiting competition and innovation. |
| 2 | High Production & Component Cost Volatility | Core components (e.g., precision temperature sensors, compressors, thermoelectric modules) are subject to global supply chain disruptions and price fluctuations. Combined with the need for medical-grade materials and rigorous quality testing, these factors drive high production costs, limiting affordability in emerging markets and squeezing profit margins. |
| 3 | Reimbursement Uncertainty & Cost-Conscious Healthcare Systems | Inconsistent or limited insurance reimbursement for heater-cooler devices and related procedures in many regions discourages hospital adoption. Cost-containment pressures in public healthcare systems further slow replacement cycles and new installations, constraining market growth. |
| 4 | Intense Competition & Market Saturation in Mature Regions | North America and Europe are dominated by a small number of established medical device manufacturers with strong brand recognition and large sales networks. New entrants face significant barriers to market share, including high customer switching costs and established clinical preference for legacy products. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
The report provides a detailed analysis of the market size, growth potential, and key trends for each segment. Through detailed analysis, industry players can identify profit opportunities, develop strategies for specific customer segments, and allocate resources effectively.
The Medical Heater Cooler Device market is segmented as below:
By Company
Getinge AB
LivaNova
ParaTherm
Spectrum Medical
SENKO MEDICAL INSTRUMENT MFG.
Medtronic
Terumo
Segment by Type
Fixed-type
Mobile-type
Segment by Application
Heart Surgery
Lung Surgery
Other
Each chapter of the report provides detailed information for readers to further understand the Medical Heater Cooler Device market:
Chapter 1: Introduces the report scope of the Medical Heater Cooler Device report, global total market size (valve, volume and price). This chapter also provides the market dynamics, latest developments of the market, the driving factors and restrictive factors of the market, the challenges and risks faced by manufacturers in the industry, and the analysis of relevant policies in the industry. (2021-2032)
Chapter 2: Detailed analysis of Medical Heater Cooler Device manufacturers competitive landscape, price, sales and revenue market share, latest development plan, merger, and acquisition information, etc. (2021-2026)
Chapter 3: Provides the analysis of various Medical Heater Cooler Device market segments by Type, covering the market size and development potential of each market segment, to help readers find the blue ocean market in different market segments. (2021-2032)
Chapter 4: Provides the analysis of various market segments by Application, covering the market size and development potential of each market segment, to help readers find the blue ocean market in different downstream markets.(2021-2032)
Chapter 5: Sales, revenue of Medical Heater Cooler Device in regional level. It provides a quantitative analysis of the market size and development potential of each region and introduces the market development, future development prospects, market space, and market size of each country in the world..(2021-2032)
Chapter 6: Sales, revenue of Medical Heater Cooler Device in country level. It provides sigmate data by Type, and by Application for each country/region.(2021-2032)
Chapter 7: Provides profiles of key players, introducing the basic situation of the main companies in the market in detail, including product sales, revenue, price, gross margin, product introduction, recent development, etc. (2021-2026)
Chapter 8: Analysis of industrial chain, including the upstream and downstream of the industry.
Chapter 9: Conclusion.
Benefits of purchasing QYResearch report:
Competitive Analysis: QYResearch provides in-depth Medical Heater Cooler Device competitive analysis, including information on key company profiles, new entrants, acquisitions, mergers, large market shear, opportunities, and challenges. These analyses provide clients with a comprehensive understanding of market conditions and competitive dynamics, enabling them to develop effective market strategies and maintain their competitive edge.
Industry Analysis: QYResearch provides Medical Heater Cooler Device comprehensive industry data and trend analysis, including raw material analysis, market application analysis, product type analysis, market demand analysis, market supply analysis, downstream market analysis, and supply chain analysis.
and trend analysis. These analyses help clients understand the direction of industry development and make informed business decisions.
Market Size: QYResearch provides Medical Heater Cooler Device market size analysis, including capacity, production, sales, production value, price, cost, and profit analysis. This data helps clients understand market size and development potential, and is an important reference for business development.
Other relevant reports of QYResearch:
Global Medical Heater Cooler Device Market Outlook, In‑Depth Analysis & Forecast to 2031
Global Medical Heater Cooler Device Sales Market Report, Competitive Analysis and Regional Opportunities 2025-2031
Global Medical Heater Cooler Device Market Research Report 2025
About Us:
QYResearch founded in California, USA in 2007, which is a leading global market research and consulting company. Our primary business include market research reports, custom reports, commissioned research, IPO consultancy, business plans, etc. With over 19 years of experience and a dedicated research team, we are well placed to provide useful information and data for your business, and we have established offices in 7 countries (include United States, Germany, Switzerland, Japan, Korea, China and India) and business partners in over 30 countries. We have provided industrial information services to more than 60,000 companies in over the world.
Contact Us:
If you have any queries regarding this report or if you would like further information, please contact us:
QY Research Inc.
Add: 17890 Castleton Street Suite 369 City of Industry CA 91748 United States
EN: https://www.qyresearch.com
Email: global@qyresearch.com
Tel: 001-626-842-1666(US)
JP: https://www.qyresearch.co.jp









