Fluid Tapping AFM Probe Market Summary
A Fluid Tapping AFM Probe is a specialized atomic force microscopy (AFM) cantilever designed for intermittent contact (tapping mode) imaging in liquid environments. Unlike standard tapping probes used in air, fluid tapping probes are optimized for operation in aqueous or buffer solutions, where damping effects and viscous drag are significantly higher. These probes typically feature a shorter cantilever with higher stiffness and a resonant frequency suitable for liquid immersion, allowing them to oscillate reliably while maintaining gentle, controlled interaction with soft or biological samples. Fluid tapping AFM probes are widely used in biophysics, molecular biology, and nanomedicine, particularly for imaging live cells, DNA, proteins, and hydrated biomaterials at nanoscale resolution under near-physiological conditions. Their precise control over tip-sample interaction enables high-resolution topographical imaging with minimal damage to delicate samples.
The current market for Fluid Tapping AFM Probes is growing steadily, driven by increasing demand for nanoscale imaging of delicate samples in liquid environments, particularly in biophysics, molecular biology and nanomedicine, as these probes are optimized to minimize sample damage while maintaining high resolution in aqueous or buffer solutions, with a concentrated competitive landscape dominated by specialized manufacturers and gradual penetration into academic research and biopharmaceutical development.
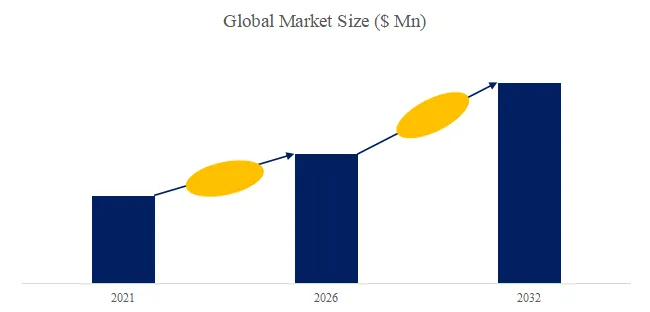
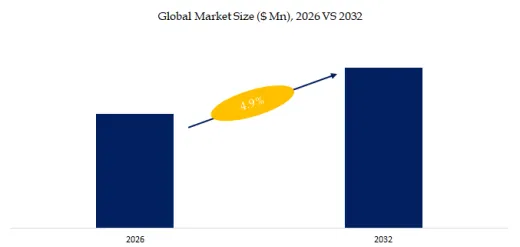
According to the new market research report “Global Fluid Tapping AFM Probe Market Report 2026-2032″, published by QYResearch, the global market for Fluid Tapping AFM Probe was valued at US$ 62.3 million in the year 2025 and is projected to reach a revised size of US$ 88.1 million by 2032, growing at a CAGR of 4.9 % during the forecast period 2026-2032.
Figure00001. Global Fluid Tapping AFM Probe Market Size (US$ Million), 2026 VS 2032
Above data is based on report from QYResearch: Global Fluid Tapping AFM Probe Market Report 2026-2032(published in 2026). If you need the latest data, plaese contact QYResearch.
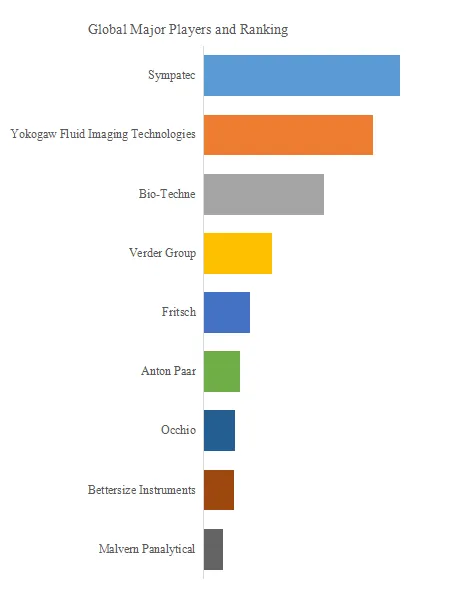
Figure00002. Global Fluid Tapping AFM Probe Top 6 Players Ranking and Market Share (Ranking is based on the revenue of 2025, continually updated)
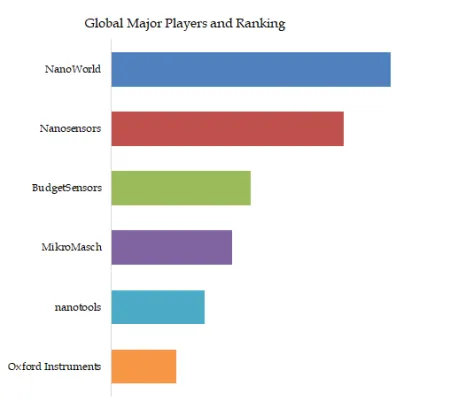
Above data is based on report from QYResearch: Global Fluid Tapping AFM Probe Market Report 2026-2032 (published in 2026). If you need the latest data, plaese contact QYResearch.
Table 1. Fluid Tapping AFM Probe Industry Chain Analysis
| Item | Description | |
| Upstream | Core Component Suppliers | Provide cantilevers, tips, magnetic coatings and conductive materials that determine the imaging resolution and stability of fluid tapping AFM probes. |
| Raw Material Providers | Supply high-purity silicon, silicon nitride, metal films and protective coatings required for probe component manufacturing. | |
| Technology R&D Institutions | Engage in research on micro-nano processing, tip sharpening and surface modification technologies to support upstream innovation. | |
| Midstream | Probe Manufacturing Enterprises | Process raw materials and assemble components to produce fluid tapping AFM probes, focusing on precision, durability and liquid compatibility. |
| Customization & Calibration Providers | Adjust probe specifications, tip geometry and cantilever stiffness to meet specific liquid-imaging research needs. | |
| Quality Inspection Bodies | Conduct tests on probe resolution, resonant frequency and stability in liquid environments to comply with nanoscale characterization standards. | |
| Downstream | Terminal Application Entities | Include academic research laboratories, biopharmaceutical companies and materials science institutions that use probes for liquid-environment nanoscale imaging. |
| Distribution Channels | Deliver products through nanotechnology reagent distributors, AFM equipment suppliers and specialized scientific instrument dealers. | |
| After-Sales & Technical Services | Offer probe usage guidance, calibration support and troubleshooting to ensure optimal performance in liquid imaging workflows. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 2. Fluid Tapping AFM Probe Industry Development Trends
| Development Trends | Description | |
| 1 | Structural and performance optimization | Fluid tapping AFM probes are continuously improved in structural design, with optimized cantilever length, stiffness and resonant frequency to reduce hydrodynamic interference in liquid environments, enhance imaging resolution and minimize damage to delicate samples. |
| 2 | Multifunctional integration | Probes are integrated with additional detection capabilities beyond basic imaging, enabling simultaneous characterization of sample topography and other physical or chemical properties to meet diverse research needs in nanoscale analysis. |
| 3 | Compatibility with automated systems | Products are designed to better match high-throughput and automated AFM platforms, supporting standardized and efficient experimental workflows and adapting to the growing demand for automated nanoscale research. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 3. Fluid Tapping AFM Probe Industry Development Opportunities
| Development Opportunities | Description | |
| 1 | Expansion of downstream research fields | The rapid development of biophysics, molecular biology, nanomedicine and materials science drives sustained demand for fluid tapping AFM probes, as they are essential for imaging biological samples and delicate materials in near-physiological or liquid environments. |
| 2 | Growing demand for high-precision liquid imaging | The increasing focus on nanoscale characterization of samples in their native liquid environments (such as living cells and hydrated materials) boosts the adoption of fluid tapping AFM probes over traditional air-based probes. |
| 3 | Advancements in AFM technology | The continuous upgrading of AFM systems, including higher precision and automation, drives the matching demand for high-performance fluid tapping AFM probes, promoting the upgrading and popularization of probe products. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Table 4. Fluid Tapping AFM Probe Obstacles/Challenges to Industry Development
| Obstacles/Challenges | Description | |
| 1 | High technical and manufacturing barriers | The production of fluid tapping AFM probes involves sophisticated micro-nano processing, surface modification and precision calibration technologies, requiring large-scale R&D investment and specialized expertise, forming high entry barriers for new enterprises. |
| 2 | High production and operational costs | The use of high-purity raw materials, complex manufacturing processes and strict quality control requirements lead to high production costs, while the limited lifespan of probes adds to operational costs for users, restricting market penetration in cost-sensitive segments. |
| 3 | Intense market competition and concentration | The market is dominated by a few specialized manufacturers with strong technical and brand advantages, leading to fierce competition; small and medium-sized enterprises face difficulties in breaking through due to lack of technical accumulation and brand recognition. |
Source: Secondary Sources, Press Releases, Expert Interviews and QYResearch, 2026
Future trends will focus on enhanced structural design to improve stability and reduce hydrodynamic interference in fluid, the development of multifunctional probes integrated with additional detection capabilities for simultaneous property mapping, and deeper compatibility with automated and high-throughput AFM systems to streamline workflows, alongside advancements in material and coating technologies to extend probe lifespan and enhance performance in complex liquid-based research scenarios.
About The Authors
| Bai Lili – Electronics Industry Analyst
bailili@qyresearch.com
Focusing on the electronics and communications field for a long time, she has observed, followed up and researched on various links in the industry chain, such as semiconductors, consumer electronics, home appliances, wired communications, wireless communication systems, Internet of Things (IoT) and smart home for a long time. She has rich experience in industry research and has completed many successful cases. |
||
About QYResearch
QYResearch founded in California, USA in 2007.It is a leading global market research and consulting company. With over 17 years’ experience and professional research team in various cities over the world QY Research focuses on management consulting, database and seminar services, IPO consulting, industry chain research and customized research to help our clients in providing non-linear revenue model and make them successful. We are globally recognized for our expansive portfolio of services, good corporate citizenship, and our strong commitment to sustainability. Up to now, we have cooperated with more than 60,000 clients across five continents. Let’s work closely with you and build a bold and better future.
QYResearch is a world-renowned large-scale consulting company. The industry covers various high-tech industry chain market segments, spanning the semiconductor industry chain (semiconductor equipment and parts, semiconductor materials, ICs, Foundry, packaging and testing, discrete devices, sensors, optoelectronic devices), photovoltaic industry chain (equipment, cells, modules, auxiliary material brackets, inverters, power station terminals), new energy automobile industry chain (batteries and materials, auto parts, batteries, motors, electronic control, automotive semiconductors, etc.), communication industry chain (communication system equipment, terminal equipment, electronic components, RF front-end, optical modules, 4G/5G/6G, broadband, IoT, digital economy, AI), advanced materials industry Chain (metal materials, polymer materials, ceramic materials, nano materials, etc.), machinery manufacturing industry chain (CNC machine tools, construction machinery, electrical machinery, 3C automation, industrial robots, lasers, industrial control, drones), food, beverages and pharmaceuticals, medical equipment, agriculture, etc.
About Us:
QYResearch founded in California, USA in 2007, which is a leading global market research and consulting company. Our primary business include market research reports, custom reports, commissioned research, IPO consultancy, business plans, etc. With over 18 years of experience and a dedicated research team, we are well placed to provide useful information and data for your business, and we have established offices in 7 countries (include United States, Germany, Switzerland, Japan, Korea, China and India) and business partners in over 30 countries. We have provided industrial information services to more than 60,000 companies in over the world.
Contact Us:
If you have any queries regarding this report or if you would like further information, please contact us:
QY Research Inc.
Add: 17890 Castleton Street Suite 369 City of Industry CA 91748 United States
EN: https://www.qyresearch.com
Email: global@qyresearch.com
Tel: 001-626-842-1666(US)
JP: https://www.qyresearch.co.jp